🧪 Periodic Table of Elements – Easy Explanation for Students
📌 Introduction
The Periodic Table of Elements is one of the most important topics in chemistry. Every student preparing for exams like JEE, NEET, and CBSE must understand it clearly.
The periodic table helps us to organize all chemical elements in a simple way. It also helps us to predict properties of elements.
In this blog, we will learn the periodic table in very easy language.
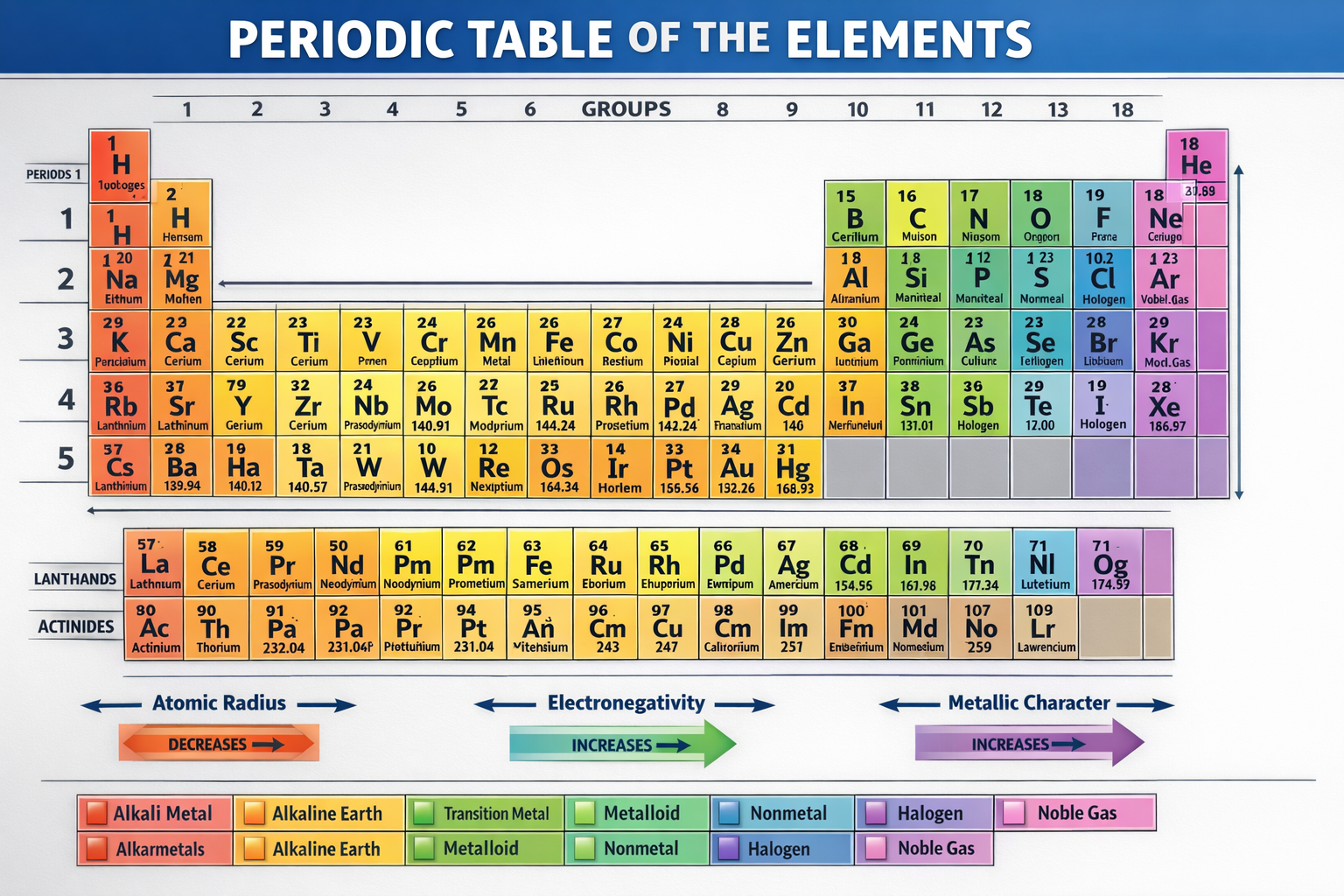
📊 What is the Periodic Table?
The periodic table is a chart of all known elements.
- Elements are arranged by atomic number
- Atomic number = number of protons
- Elements with similar properties are placed together
👉 Example:
- Sodium (Na) and Potassium (K) are in the same group
- Both are highly reactive metals
Grp→ 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18
P1: H He
P2: Li Be B C N O F Ne
P3: Na Mg Al Si P S Cl Ar
P4: K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr
P5: Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe
P6: Cs Ba La* Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn
P7: Fr Ra Ac* Rf Db Sg Bh Hs Mt Ds Rg Cn Nh Fl Mc Lv Ts Og
Lanthanides: La → Lu (57–71)
Actinides: Ac → Lr (89–103)
🧱 Structure of Periodic Table
The periodic table has:
🔹 Periods (Rows)
- Total 7 periods
- Horizontal rows
- Properties change gradually across a period
🔹 Groups (Columns)
- Total 18 groups
- Vertical columns
- Elements in the same group have similar properties
🧩 Types of Elements
The periodic table has different types of elements:
1️⃣ Alkali Metals (Group 1)
- Very reactive metals
- Soft and shiny
- Example: Sodium (Na), Potassium (K)
2️⃣ Alkaline Earth Metals (Group 2)
- Less reactive than alkali metals
- Example: Magnesium (Mg), Calcium (Ca)
3️⃣ Transition Metals (Group 3–12)
- Strong and hard metals
- Used in industries
- Example: Iron (Fe), Copper (Cu)
4️⃣ Halogens (Group 17)
- Very reactive non-metals
- Form salts
- Example: Chlorine (Cl), Fluorine (F)
5️⃣ Noble Gases (Group 18)
- Very stable
- Do not react easily
- Example: Neon (Ne), Argon (Ar)
🧪 Blocks in Periodic Table
The periodic table is divided into blocks:
- s-block → Groups 1 and 2
- p-block → Groups 13 to 18
- d-block → Transition metals
- f-block → Lanthanides and Actinides
👉 This classification helps students understand electron configuration easily.
📈 Important Periodic Trends
Periodic trends are very important for exams.
🔹 1. Atomic Radius
- Size of an atom
✔ Decreases from left to right
✔ Increases from top to bottom
👉 Reason:
- Nuclear charge increases across a period
🔹 2. Electronegativity
- Ability to attract electrons
✔ Increases across a period
✔ Decreases down a group
👉 Fluorine is the most electronegative element.
🔹 3. Metallic Character
- Tendency to lose electrons
✔ Decreases across a period
✔ Increases down a group
👉 Metals are mostly on the left side.
🔹 4. Ionization Energy
- Energy required to remove an electron
✔ Increases across
✔ Decreases down
🧭 Position of Elements
- Left side → Metals
- Right side → Non-metals
- Middle → Transition metals
- Zig-zag line → Metalloids
⭐ Special Series
🔸 Lanthanides (57–71)
- Called rare earth elements
- Used in electronics
🔸 Actinides (89–103)
- Radioactive elements
- Example: Uranium (U)
🎯 Why Periodic Table is Important?
The periodic table is useful because:
- Helps to understand element properties
- Helps to predict chemical reactions
- Important for competitive exams
- Used in real-life applications
🧠 Easy Memory Tips
- Group 1 → Highly reactive metals
- Group 18 → Stable gases
- Left → Metals
- Right → Non-metals
👉 Shortcut:
“Left = Lose electrons, Right = Gain electrons”
📚 Conclusion
The periodic table is the heart of chemistry. If you understand it well, many topics become easy.
Start by learning:
- Groups and periods
- Types of elements
- Important trends
With practice, you will master it easily.
🚀 Final Tip for Students
- Revise the periodic table daily
- Use charts and diagrams
- Practice questions regularly



